-
Publish Your Research/Review Articles in our High Quality Journal for just USD $99*+Taxes( *T&C Apply)
Offer Ends On
Drishya Mukherjee, Ananya Bera, Ramanuj Mukherjee, Siddhartha Chatterjee and Amitava Chatterjee*
Corresponding Author: Amitava Chatterjee, Calcutta Fertility Mission (A Unit of Kashyap Initiative Pvt. Ltd.), Kolkata, India.
Received: January 15, 2025 ; Revised: February 15, 2025 ; Accepted: February 18, 2025 ; Available Online: March 12, 2025
Citation: Mukherjee D, Bera A & Mukherjee R, Chatterjee S & Chatterjee A. (2025) Salivary MMPs (Matrixmetalloproteinases) in the Diagnosis of Human Breast Cancer. J Can Sci Res Ther, 5(1): 1-3.
Copyrights: ©2025 Mukherjee D, Bera A & Mukherjee R, Chatterjee S & Chatterjee A. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Views & Citations
Likes & Shares
MMPs and TIMP-2 plays important role in cancer cell invasion. human saliva is a biological fluid of varying diagnostic potential with several advantages of disease diagnosis and prognosis. The Matrixmetallo Proteases (MMPs) is a family of proteases involved in extra cellular matrix remodeling. The activity of MMPs has been implicated in a number of important normal and pathologic processes. Tumor growth, metastasis and angiogenesis are associated with this event.
Keywords: Human female breast cancer, Matrix metalloproteinases (MMPs), Saliva
INTRODUCTION
The MMPs family of extracellular proteases has long been associated with cancer metastasis by virtue of them to degrade all the components of ECM. MMPs and TIMP-2 plays important role in cancer cell invasion. The MMPs are involved in extracellular matrix remodeling [1-5]. Targeting of these enzymes as diagnostic and prognostic agents for detection of human cancers are well thought. Salivary markers have been reported for oral cancer detection [1,2]. A review on salivary proteomics and genomics has been reported [3]. The Matrix metalloproteinases (MMPs) is a family of proteases involved in extra cellular matrix remodeling. The activity of MMPs has been implicated in a number of important normal and pathologic processes [4-8]. Tumor growth and metastasis and angiogenesis are associated with this event. The role of MMPs in Carcinogenesis is well studied [9-12]. Biomarkers are also called tumor markers for breast, there are several tumor markers like CA-125, BRCA 1 & 2, Her-2, ki-67 etc. [9]. Saliva use is safe and it does not contaminate and does not require any sophisticated method. MMPs as cancer biomarker has been studied in many cancers. MMPs shape the tumor microenvironment. The role of MMPs in cancer biology especially in migration of malignant tumor cells is well documented. MMPs are secreted by mammalian cells as zymogens [12]. MMP-2 is activated on the cell surface by MT1-MMP [13].
METHOD AND MATERIALS
1 ml of saliva were collected from 20 female breast cancer patients (Stage- iv). Saliva of different human normal females (same age group) were also collected and centrifuged at 6000 rpm for 15 min.
Method:
Gel electrophoresis (Zymography): The salivas were run in 8% SDS- PAGE impregnated with 0.1% Gelatin. The gels were run at 25mA using TRIS -GLYCIN- SDS buffer (pH 8.3). The gels were washed in 2.5 % Triton -X for 10 min. and then incubated in buffer A (Sodium Chloride-0.2 M, Calcium chloride-4.5 mM, Tris-50 mM, pH 7.4) overnight at 37°C. The Gel was stained with CoommassieBriliant Blue to develop the zymograms [10].
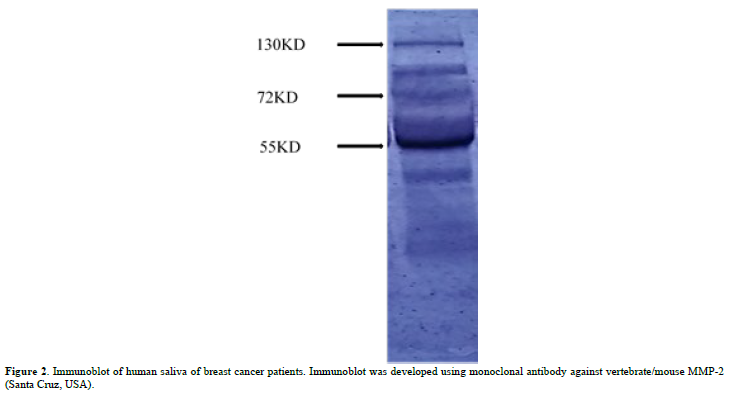
Immunoblot: The saliva sample with 0.1 % gelatin impregnated 8% gel at 25 mA. Reacted with MMP-2 monoclonal antibody (Santa Cruz, U.S.A). The bands were developed using NBT/BCIP [11]. Very clearly the 72kd band with its activated product is visible.
Protein estimation
Lowry’s method: The optical density was measured at 750 nm.
DISCUSSION
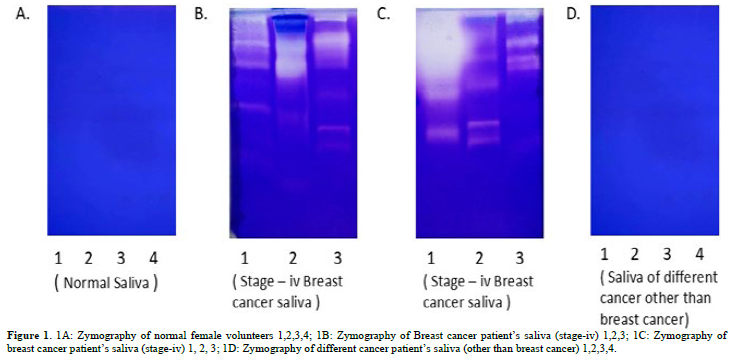
Figure 1A shows the saliva of the normal individual, which shows no MMP bands. Figures 1B & 1C shows the breast cancer salivas of stage -iv and shows number of MMP bands including MMP-9 and activated MMP-2, probably. Figure 1D shows other cancers (cervical, ovarian, liver, prostate etc.) absent of MMPs. The Figure 2 shows MMP-2 immunoblot. The 72kd MMP-2 is less and the 55kd activated product is more. The VEGF also shows that in pre-surgery cases it is more than the post-surgery cases. VEGF is a positive regulator of MMP-2 [10]. We are thinking of making it a new method for breast cancer evaluation. Work is going on. In previous communication we reported that MMP-2 in activated form is related to this event. A band at 140kd region is also visible indicating MMP-2 is complexed with other protein. The TIMP-2 said to be MMP-2 inhibitor was found to be higher in post-surgical samples as compared to pre-surgical saliva. The reason is not yet clear why MMP is related to breast cancer only. Work is going on to clear the questions. Zymograms of the pre and post-surgery saliva of the same patient shows significant differences especially in MMP-2 with activated products [10]. Recently, it has been shown that MMP-2 has been involved in the invasion and metastasis [14-17].
No Files Found
Share Your Publication :